Studying Rats May Make It Easier to Identify, Test Therapies for Fabry Eye Problems
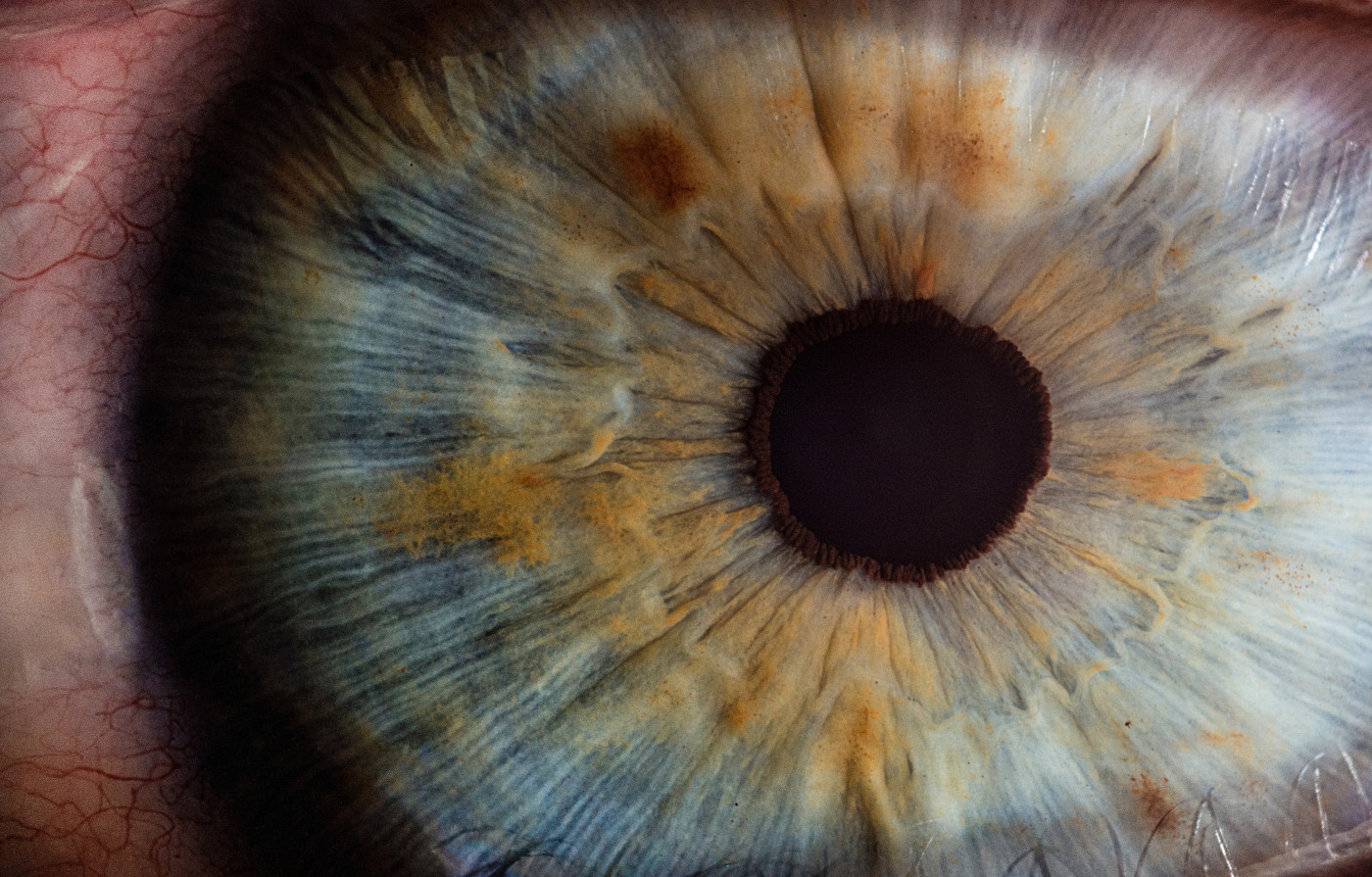
Rats that mimic Fabry disease develop eye symptoms like those observed in human patients. These animals can be used to test current and future therapies for eye problems resulting from the disease, as well as provide new insights into their underlying biology.
The study with that finding, “Rats deficient in α-galactosidase A develop ocular manifestations of Fabry disease,” was published recently in the journal Scientific Reports.
Fabry disease is a rare genetic disorder that prevents the body from making an enzyme called alpha-galactosidase A (α-Gal A). This enzyme is responsible for breaking down complex fat-sugar molecules, specifically globotriaosylceramide (Gb3 or GL-3) and related molecules. As a result, these compounds build up in cells throughout the body, particularly in cells lining blood vessels in the skin and cells in the kidneys, heart, and nervous system
The disease has a wide range of symptoms, including chronic pain, clusters of small, dark red spots in various locations on the skin, ringing in the ears, and hearing loss. Fabry also can have life-threatening symptoms, such as heart attack, stroke, and kidney disease.
Eye manifestations also are one of the most common and earliest signs of the disease, serving as important diagnostic markers. The two most frequent problems include cornea verticillata (circle-like opacities in the cornea) and cataracts (a clouding, or opacity of the lens).
Some patients also may have tortuous, or twisted, vessels in the conjunctiva (mucous membrane that lines the eyelid and covers the visible portion of the eyeball), retina (the layer of nervous tissue in the back of the eye responsible for sensing light), or in the choroid (part of the middle coat of the eye).
While there is evidence that ocular signs correlate well with disease severity in patients, it is unknown whether eye opacities stabilize or decrease with therapy, namely enzyme replacement therapy (ERT).
Studying eye involvement using animal models of Fabry disease could be useful, but to date no studies have evaluated if these models develop eye problems identical to patients.
With that in mind, researchers carefully examined if rat models of Fabry also had signs of eye problems. The team used a lab-made rat model of Fabry, which is deficient for alpha-galactosidase A and “which demonstrates pain, cardiac, and renal phenotypes [traits] commonly observed in patients.”
They found that Fabry rats were more susceptible to develop opaque corneas and cataracts, as do Fabry patients. However, unlike humans, they had no overt twisting of retinal vessels.
Fabry rats also accumulated fat-sugar molecules that normally are broken down by alpha-galactosidase A (galactosyl glycosphingolipids) in several parts of the eye — keratocytes (specialized cells of the cornea), lens fibers, and cells lining the vessels of the retina.
“To our knowledge, this is the first study that extensively characterizes ocular phenotypes [traits] in an animal model of Fabry disease,” the researchers wrote.
This finding may “have tremendous potential” to inform future therapy studies, the researchers stressed.
Fabry rats can be used to understand if eye problems can regress with current or future therapies, or what the optimal age would be to begin therapy to obtain maximum benefit. Also, they provide a model through which the biology of eye disease in Fabry can be investigated.






